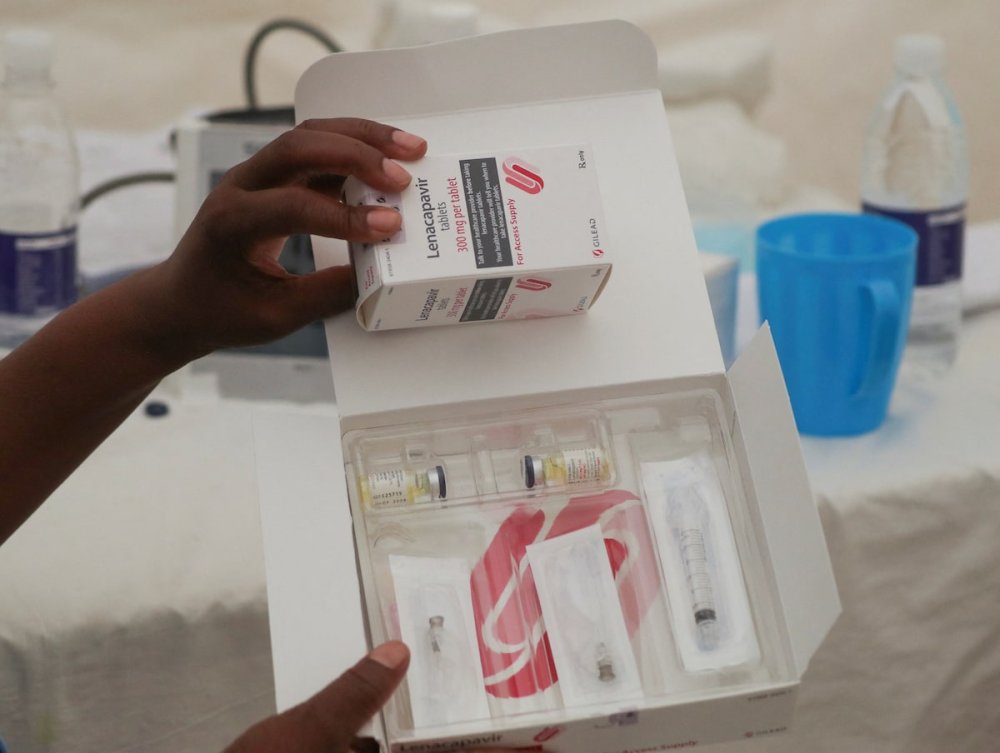
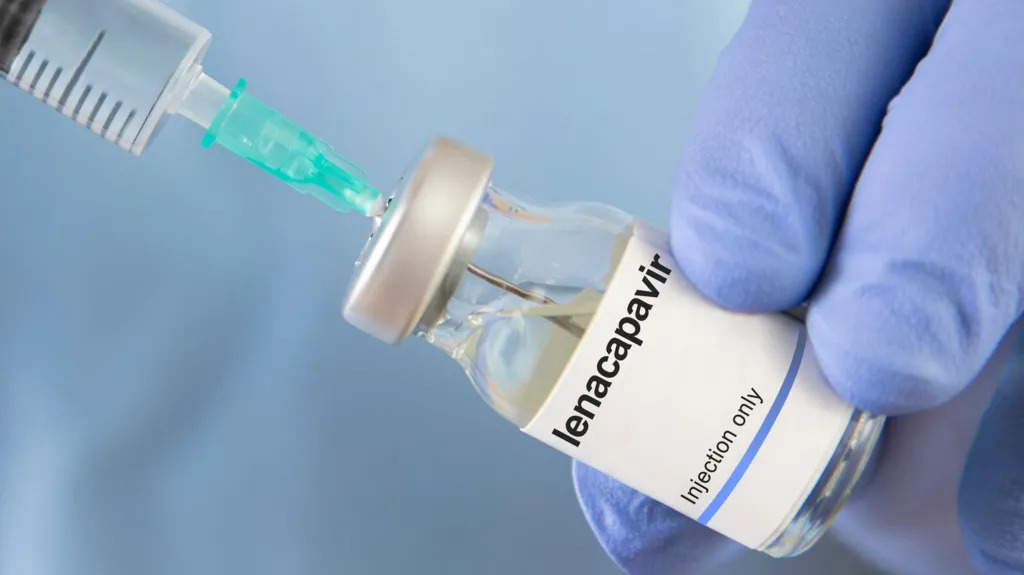
The announcement, made on April 14, 2026, comes as the two partners scale up their commitment to increase access to HIV pre-exposure prophylaxis (PrEP) using lenacapavir, a twice-yearly injectable that has shown near 100% effectiveness in clinical trials.
Under the expanded initiative, an additional 1 million people are expected to be reached over the next three years, bringing the total target to 3 million people by 2028.
Lenacapavir is designed as a long-acting alternative to daily oral PrEP, offering a discreet and highly effective option for HIV prevention. Health experts say it could transform prevention efforts, particularly among populations that face challenges with daily medication adherence.
“This expanded commitment in partnership with the United States reflects both the scale of the opportunity and the urgency of the moment,” said Peter Sands, Executive Director of the Global Fund. He noted that long-acting prevention tools could help overcome long-standing barriers that have limited uptake of HIV prevention services.
Rwanda joins Benin, Botswana, Dominican Republic, Fiji, Georgia, Haiti, Honduras, Indonesia, Morocco, Papua New Guinea and Thailand as new countries supported for introduction of the injectable prevention tool.
The rollout builds on earlier distribution to nine African countries, including Kenya, Uganda, South Africa, Nigeria, Zambia and Zimbabwe, where early data shows strong uptake among priority groups such as adolescent girls and young women, pregnant and breastfeeding women, and individuals accessing PrEP for the first time.
According to the Global Fund, the expanding interest from countries reflects growing demand for more user-friendly HIV prevention methods that reduce reliance on daily pills. Early implementation data suggests that injectable PrEP could significantly improve access among people who have not previously used prevention services.
The initiative is also being supported through voluntary licensing agreements by Gilead Sciences, allowing manufacturers to produce more affordable generic versions of lenacapavir. This is expected to expand supply in the coming years, alongside ongoing shipments of the original product.
Global Fund officials say the priority now is ensuring rapid but responsible rollout, with strong involvement of community-led organisations in building awareness, trust, and uptake in high-risk populations.
“The level of country demand we are seeing is both encouraging and urgent,” said Mark Edington, Head of Grant Management at the Global Fund. “Countries recognise the transformative potential of LEN to reach populations that have not been adequately served by existing tools.”
The Global Fund emphasised that it is working closely with governments and partners to align implementation with national HIV strategies and ensure equitable access as the program scales up.
The expanded initiative is expected to significantly strengthen global HIV prevention efforts, with Rwanda among the countries set to benefit from the introduction of one of the most promising new prevention technologies in recent years.
Leave a Reply