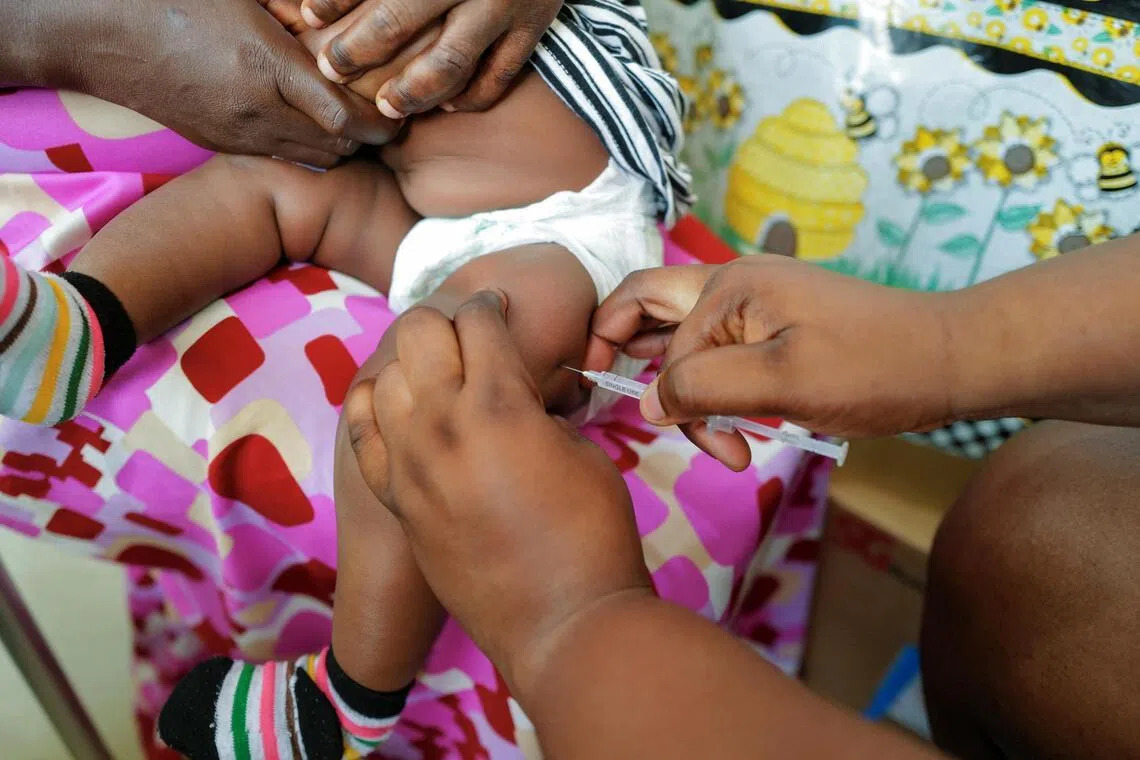
The newly approved medicine, artemether-lumefantrine, is tailored for babies weighing between two and five kilograms. Previously, infants diagnosed with malaria were typically treated with drugs formulated for older children, creating challenges in achieving accurate dosing and increasing the risk of side effects.
WHO’s approval, announced on April 24, confirms that the treatment meets international standards for safety and effectiveness. The decision also clears the way for governments and health agencies to procure and distribute the drug more widely, potentially improving access to life-saving care.
Health experts say the move could help close a long-standing treatment gap, particularly in Africa, where an estimated 30 million babies are born each year in malaria-endemic regions.
Speaking on the development, WHO Director-General Tedros Adhanom Ghebreyesus said recent advances are helping to shift the trajectory of the global malaria fight. He highlighted progress in vaccines, diagnostics, mosquito control tools and medicines, including those adapted for the youngest patients.
“For centuries, malaria has stolen children from their parents, and health, wealth and hope from communities,” he said. “But today, the story is changing. New vaccines, diagnostic tests, next-generation mosquito nets and effective medicines are helping to turn the tide.”
In addition to the new treatment, WHO has approved three rapid diagnostic tests aimed at improving malaria detection. Many existing tests identify a protein known as HRP2, but some malaria parasites have evolved to stop producing it, making infections harder to detect.
This challenge has been reported in parts of eastern Africa, including Somalia, Ethiopia, Eritrea and Djibouti, where gaps in diagnosis have led to missed cases. The newly approved tests target a different protein, pf-LDH, which is less prone to change.
Public health experts believe this will significantly improve diagnostic accuracy in regions where current testing methods are failing. WHO recommends that countries switch to the new tests if more than five percent of malaria cases go undetected.
In Rwanda, the arrival of infant-specific treatments coincides with a period of intensified surveillance. According to recent data from the Rwanda Biomedical Centre (RBC), malaria incidence in the country rose to 76 cases per 1,000 people in the 2024/25 fiscal year, up from 45 the previous year. Total malaria cases reached 1,131,314 during that peak, though early 2026 reports show a promising dip toward 928,000 cases.
Despite these advances, malaria remains a major global health challenge. According to WHO’s latest World Malaria Report, there were approximately 282 million cases and 610,000 deaths in 2024, an increase of about 9 million cases compared to the previous year.
While some countries have made notable progress in reducing or eliminating the disease, efforts have slowed in many regions due to factors such as drug and insecticide resistance, weak diagnostic systems and declining funding.
The approval of a newborn-specific treatment is seen as a critical step in addressing these challenges and strengthening the global response to malaria, particularly among those most at risk.
Leave a Reply